Topiramate XR
There are 2 currently approved Brand name extended release versions of topiramate XR: Qudexy XR and Trokendi XR. These brand names are used in the United States for the extended-release form of the seizure medicine topiramate.
Qudexy XR and the Topiramate Extended-Release Capsules generic are approved to use:
- Alone (as monotherapy) in children 2 years of age and older and in adults with partial seizures and primary generalized tonic clonic seizures.
- With other seizure medicines (as adjunctive or add-on treatment) in children 2 years of age and older and adults with partial seizures, primary generalized tonic clonic seizures and seizures associated with Lennox-Gastaut syndrome (LGS).
Qudexy XR is marketed in the United States by Upsher-Smith. The name or appearance may differ in other countries. The name or appearance may differ in other countries. The following descriptions apply to the US versions: Qudexy XR is available in 25 mg, 50 mg, 100 mg, 150 mg, and 200 mg extended release capsules.
A generic version of Qudexy XR, Topiramate Extended-Release Capsules, is made by the same manufacturer of Qudexy XR (Upsher-Smith). This generic form has the same active and inactive ingedients as Qudexy XR.
Trokendi XR is approved to use:
- Alone (as monotherapy) in children 10 years of age and older and in adults with partial seizures and primary generalized tonic clonic seizures.
- With other seizure medicines (as adjunctive or add-on treatment) in children 6 years of age and older and in adults with partial seizures, primary generalized tonic clonic seizures and seizures associated with LGS.
Trokendi XR is marketed in the United States by Supernus. The name or appearance may differ in other countries. The dose (measured in milligrams, abbreviated "mg") will usually be the same. These descriptions apply to the US versions: Trokendi XR is available in 25 mg, 50 mg, 100 mg and 200 mg extended release capsules.
The dose of Qudexy XR or Trokendi XR will vary depending on a number of different factors including age, weight and your seizure type and whether you are taking this medication as your only drug to control seizures or whether it is being added on to another medication. Typical starting dose of the medication is 25-50 mg once daily. The typical recommended dose in adults is 400 milligrams once daily.
Follow the directions on how much or what dose to take that are given by your prescribing health care provider. Call if you have any questions.
- Usually, dosing is started slowly. You may be asked to start taking one 50-milligram tablet once a day.
- After a while, the doctor may gradually increase the dosage to get better control of your seizures.
- Because topiramate is generally taken two times a day, many people help themselves to remember by taking it with breakfast and dinner.
- Ask the doctor what to do if you forget to take a dose.
Be careful if the doctor writes a new prescription using a different kind of pill. For example, if you've been using 25-mg tablets and the new prescription is for 100-mg tablets, be careful to use the correct number of pills and dose. Don't automatically continue to use the same number of pills as before. Make sure you know what size tablet has been prescribed.
Updated: 16/10/2023
Brand Name(s)
Qudexy XR, Trokendi XR
Used to Treat
Forms
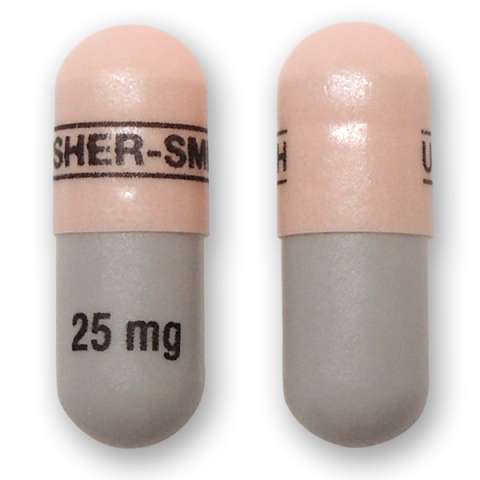
25 mg
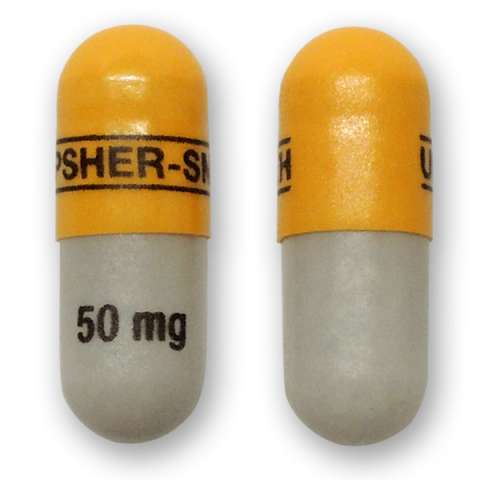
50 mg
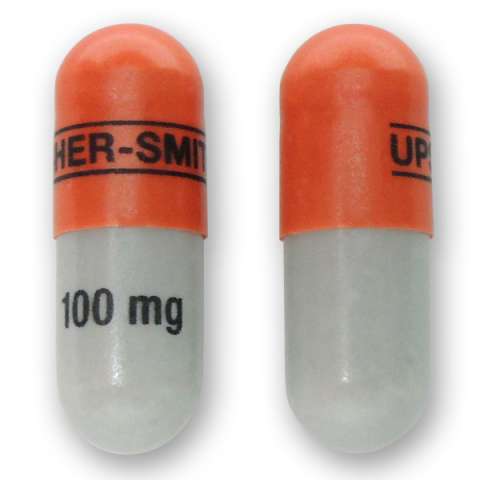
100 mg
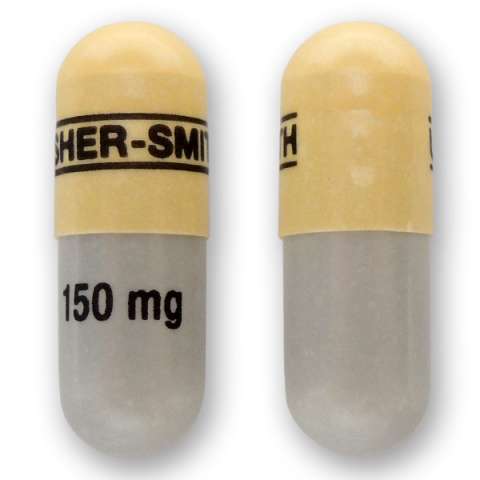
150 mg

200 mg

25 mg: light green opaque body/yellow opaque cap (printed “SPN” on the cap, “25” on the body)

50 mg: light green opaque body/orange opaque cap (printed “SPN” on the cap, “50” on the body)

100 mg: green opaque body/blue opaque cap (printed “SPN” on the cap, “100” on the body)

200 mg: pink opaque body/blue opaque cap (printed “SPN” on the cap, “200” on the body)
Package Insert
Frequently Asked Questions
How to take and store Topiramate XR?
How to take Topiramate XR
The tablet or capsule forms of both brands and the generic can be swallowed whole with a glass of water. Don't bite them as they may have a bitter taste.
All strengths of Qudexy™ XR and its authorized generic form may be swallowed whole or given by carefully opening the capsule and sprinkling the entire contents on a spoonful of soft food. Sprinkling the drug onto food may be helpful for children or people have have problems swallowing whole capsules or tablets.
Be sure to use only the amount of Topiramate XR that your prescribing health care professional recommends. If you think you've taken one or two extra tablets, call your doctor for advice. For a larger overdose, call your local poison control center or emergency room right away, unless you have special directions from the doctor.
Don't stop taking Qudexy XR or Trokendi XR or change the amount you take without talking to your doctor first. Stopping any seizure medicine all at once can cause serious problems.
How to store Topirmate XR
Both the tablets and capsules should be stored at room temperature (59° to 86°F or 15° to 30°C), in a tightly sealed container protected from moisture and humidity. Be especially careful to protect the sprinkle capsules from heat and store below 77°F (25°C). If you pick up the prescription in warm weather, don't leave them in the car while you run other errands!
What if I forget?
If you forget a dose, take it as soon as you remember. If it is almost time for the next dose, delay that dose for a few hours instead of taking two doses very close together. Talk to your doctor for specific instructions on how to make it up. Do not double up or take extra medicine, unless instructed to do so by your doctor.
Do your best to follow the doctor's directions. If you forget doses often, it may be a good idea to get a special pillbox or watch with an alarm to remind you.
Taking the right amount of seizure medicine on time every single day is the most important step in preventing seizures!
How does Topiramate XR affect the brain?
Brain cells need to work (fire) at a certain rate to function normally. During a seizure, brain cells are forced to work much more rapidly than normal. Qudexy XR helps prevent brain cells from working as fast as a seizure requires them to. In this way, seizures can be stopped when they are just beginning.
How does the body digest Topiramate XR?
After medicine is swallowed, it must be absorbed into the blood so it can move throughout the body. The process of absorbing, digesting, and excreting a medicine or food is called metabolism. The way the body metabolizes a particular medicine affects how often it must be taken. It also determines whether it will interact with other medicines or be affected by liver disease or kidney disease.
Like many other medicines, Qudexy XR is broken down (digested) in the liver. People with liver disease must be cautious about taking it. Other people also need to be careful if they take other medicines that are digested in the liver. The metabolism of each medicine may be affected, changing how quickly it works and leaves the body.
Qudexy XR is eliminated from the body through the kidneys.
How well does the Topiramate XR work?
Doctors have studied large numbers of people with partial seizures, generalized tonic-clonic (grand mal) seizures, or seizures from Lennox-Gastaut syndrome to find out how well Qudexy XR or Trokendi XR works.
Studies have shown that Qudexy XR and Trokendi XR works well alone(monotherapy)and when added to other seizure medicines. Qudexy XR and Trokendi XR are not a perfect add-on seizure medicine for everyone, however. Sometimes people must try a series of combinations before finding what is best for them. Some other medicines affect how Qudexy XR or Trokendi XR is eliminated from the body, so the dosages may need to be changed with different combinations.
What are the most common side effects of Topiramate XR?
Most people who take Qudexy XR or Trokendi XR (topiramate) have little or no problem with side effects. Those with problems most often complain of:
- fatigue or drowsiness
- difficulty with concentration
- difficulty finding the right word (word retrieval)
- confusion
- dizziness
- unsteadiness
- a feeling of pins and needles, usually in the tips of the fingers and toes
- loss of appetite and weight loss
- nervousness
- depression
- difficulty with memory
Most of these problems are more common when higher doses of Qudexy XR are taken.
A few people do experience psychiatric disorders when they take Qudexy XR.
If you notice any of these problems, call the doctor's office. Sometimes the doctor can help by changing the amount of Qudexy XR or Trokendi XR you take or how you take it. Don't stop taking Qudexy XR or Trokendi XR or change the amount you take without the doctor's guidance.
The most common side effects with Qudexy XR or Trokendi XR are fatigue and sleepiness. If you've just started taking Qudexy XR or Trokendi XR or have just had your dosage increased, be careful about doing things that could be dangerous (like driving or operating machinery) until you know how it will affect you. Be especially cautious if you tend to be sensitive to medications.
Be sure to read about the serious side effects so you will be aware of a few serious problems that could arise when you take Qudexy XR or Trokendi XR These serious problems are very rare but everyone who takes this medicine should at least be aware of them.
You shouldn't stop taking Qudexy XR or Trokendi XR or any other seizure medicine without your doctor's advice.
Allergic reactions
A small percentage of people who take Qudexy XR or Trokendi XR develop a skin rash within the first few weeks of taking it. If this happens, tell the doctor or nurse right away, to be sure that it's not the beginning of a serious problem. It's rare for the rash to be serious, but don't ignore it. It's often necessary to switch to a different seizure medicine.
What are the most serious side effects of Topiramate XR?
Very few people have serious reactions to Qudexy XR or Trokendi XR. If you take it, you should be aware of them, however, so you and your family can recognize them.
Glaucoma
A very small number of people treated with Qudexy XR or Trokendi XR developed an eye condition like glaucoma. If you experience blurred vision or difficulty seeing that comes on quickly, perhaps with eye pain, notify your doctor immediately. These symptoms usually occurred during the first month of treatment. If action is taken promptly (including stopping the Qudexy XR or Trokendi XR) the symptoms will go away, but if they are not treated, permanent loss of vision could result.
Kidney stones
About 1 in 50 adults taking Qudexy XR have developed kidney stones. They are more common in men and in those who have had them before. They also may be more likely to occur if Diamox (acetazolamide) or Zonegran (zonisamide) is also taken or if the ketogenic diet (a special diet sometimes used to control seizures in children) is followed. Qudexy XR therefore should be used cautiously in people taking Diamox or Zonegran or using the ketogenic diet.
To help prevent kidney stones, drink plenty of water, juice, or other fluids (8 glasses every day for adults). Sharp pains in your side or lower back may signal the onset of a kidney stone. Consult a doctor immediately or go to the emergency room.
Acidosis
One factor that may increase the risk of kidney stones, fatigue, and other disorders is abnormally acid blood, called metabolic acidosis. Symptoms of metabolic acidosis may include tiredness, loss of appetite, irregular heartbeat, and impaired consciousness. Call your health care professional right away if these symptoms develop while taking Qudexy XR .
This effect has been found to some extent in up to two-thirds of children taking Qudexy XR and about one-third of adults. Much smaller numbers show very abnormal results on blood tests.
The doctor will probably test the blood before starting treatment with Qudexy XR and again from time to time while Qudexy XR is being used, to check for acidosis. If the results are abnormal, the doctor may recommend stopping Qudexy XR or starting treatment for acidosis to prevent any serious effects.
Inadequate sweating
Some children who take Qudexy XR may not sweat enough in hot weather, causing their body temperatures to rise, sometimes to dangerous levels. Children should be monitored in hot weather to be sure they sweat appropriately.
Bone Health
While the effect of Qudexy XR on bone health is not fully known, a person who develops metabolic acidosis can develop thinning of the bones and bone loss if the condition is not treated properly. Regular exercise and diet are very important to help keep strong bones. People should talk to their doctor before taking calcium supplements however, because extra calcium could increase the risk of kidney stones in some people who are also taking topiramate. People who have thinning of the bones may also need to take prescription medicines to treat this. Talk to your doctor about tests to check your bone health and what to do next.
A complete list of all reactions to Qudexy XR can be found in the package insert. It is important to remember that only a tiny number of people who take Qudexy XR have any of these serious problems.
If you believe that you have experienced a serious side effect from a medication, you or your physician can bring it to the attention of the FDA, through their MedWatch program, by completing an adverse event report form (http://www.fda.gov/medwatch/getforms.htm). MedWatch is the FDA's program for reporting serious reactions and problems with medical products, such as drugs and medical devices. (http://www.fda.gov/medwatch/getforms.htm.) To learn more about the Medwatch program go to: http://www.fda.gov/medwatch/report/consumer/consumer.htm
On July 10, 2008, an advisory panel was convened by the Food and Drug Administration (FDA) to review data that the FDA had previously collected from drug studies showing an association between many of the antiepileptic drugs (AEDs) and suicidal ideation and behavior, which together are called suicidality. According to the FDA’s Alert, among the patients with epilepsy in these drug studies, 1 out of 1000 people taking the placebo (inactive substance) showed suicidality compared to approximately 3.5 out of 1000 people who took an AED. The FDA advisory panel voted to accept the FDA's data at its meeting on July 10.
- Taking antiepileptic medicines may increase the risk of having suicidal thoughts or actions;
- Do not make any changes to the medication regimen without first talking with the responsible healthcare professional;
- Pay close attention to any day-to-day changes in mood, behavior and actions. These changes can happen very quickly so it is important to be mindful of any sudden differences.
- Be aware of common warning signs that might be a signal for risk of suicide. Some of these are:
- Talking or thinking about wanting to hurt yourself or end your life
- Withdrawing from friends and family
- Becoming depressed or having your depression get worse
- Becoming preoccupied with death and dying
- Giving away prized possessions
We again urge patients and families to contact their doctor before stopping an epilepsy medication because this may possibly lead to seizures and worsening of mood.
Impact of Topiramate XR on bone health
See package insert.
What else is Topiramate XR used for?
Often doctors find that medicines are useful for more than one purpose. It is legal to prescribe medicines for ""off-label uses"" even though the FDA has not formally approved such use. Common off-label uses of Qudexy XR or Trokendsi XR include:
- a type of seizure called infantile spasms (West syndrome)
- a type of seizure called myoclonic seizures
- the prevention of migraine headaches
- weight loss
- painful nerve conditions
Who should not take Topiramate XR?
The only people who definitely should not take Qudexy XR or Trokendi XR are those who are allergic to it. People with a history of kidney stones or those who need to take high doses of vitamin C or calcium supplements, however, also probably should not take Qudexy XR or Trokendsi XR because of the increased risk of kidney stones.
People who have kidney disease need to be extra cautious about taking Qudexy XR or Trokendsi XR.
Can Topiramate XR be taken with other medicines?
Sometimes one kind of medicine changes the way another kind of medicine works in the body. If two kinds of medicine affect each other, the doctor may prescribe something else or change the amount to be taken.
Qudexy XR or Trokendi XR makes birth control pills less effective, so the chances of becoming pregnant are greater. Women who use pills for birth control should talk to the doctor who prescribed them right away if they start taking Qudexy XR or Trokendi XR. The same is true for some other forms of birth control such as Depo-Provera or implants. Qudexy XR or Trokendi XR does not affect barrier types of birth control, like condoms, IUDs, and diaphragms.
Qudexy XR and Trokendi XR also affects the way the body handles phenytoin (Dilantin, Phenytek), so if you take phenytoin, ask your doctor whether you should change your dose of phenytoin.
How do other medicines affect Qudexy XR or Trokendi XR?
Your body gets rid of Qudexy XR or Trokendi XR quicker if you are also taking certain seizure medicines, such as:
- carbamazepine (Tegretol, Tegretol XR, Carbatrol)
- phenytoin (Dilantin, Phenytek)
- phenobarbital
- primidone (Mysoline)
If this applies to you, your doctor may plan to give you more Qudexy XR than you would otherwise take.
Any time one of your doctors suggests starting or stopping a prescription, be sure to ask what effect it might have on the Qudexy XR or Trokendi XR and whether the dosage should be increased or decreased.
What are the effects of Topiramate XR on Children?
Qudexy XR and Trokendi XR is used in addition to other seizure medicines to treat children with partial seizures, children with Lennox-Gastaut syndrome, and children with primary generalized tonic-clonic seizures.
Doctors figure out how much medicine to give to young children based mostly on their weight. To keep side effects at a minimum, the doctor probably will prescribe a low dose to start with and increase it slowly. Children usually start with a dose of 15 to 25 milligrams (mg) or less per day, based on a range of 1 to 3 mg per kilogram (kg, about 2.2 pounds) of body weight per day. This dose is given every night for the first week. The dose is usually then increased every week or two by adding 1 to 3 mg per kg per day (given in two divided doses) to achieve the best response. Most children do best at about 5 to 9 mg per kg per day.
If the dosage is increased slowly and carefully monitored, side effects should not be a problem for most children. The most common side effects involve problems with thinking or behavior, such as difficulty with concentration and attention or memory difficulty. These side effects may be less frequent in children than in adults. Headache, sleepiness, fatigue, and weight loss are other common side effects. Children are also less like to get the rare eye problem (glaucoma) – still, call the doctor immediately if the child experiences vision problems or eye pain.
Children should be monitored in hot weather to be sure they sweat appropriately. Qudexy XR and Trokendi XR impairs sweating in a few children, leading to high body temperatures.
If a woman takes Topiramate XR during pregnancy will it hurt the baby?
In the United States, the Food and Drug Administration (FDA) assigns each medication to a Pregnancy Category according to whether it has been proven to be harmful in pregnancy. Qudexy XR and Trokendi XR is listed in Pregnancy Category C. This indicates that caution is advised, but the benefits of the medicine may outweigh the potential risks. Studies in animals have shown some harm to the baby, but there haven't been any good studies of results in women.
Talk to your doctor or another health professional if you are pregnant or plan to become pregnant. We don't yet have enough information to be able to estimate the risk of various types of birth defects that might occur if Qudexy XR or Trokendi XRis taken during pregnancy. We also don't know enough to compare the risk with Qudexy XR or Trokendi XR to the risk with other seizure medicines. The risk of birth defects is generally higher for women who take more than one seizure medication and for women with a family history of birth defects.
Women who are capable of becoming pregnant should take at least 400 mcg (0.4 mg) of folic acid (folate) daily to help prevent a type of birth defect called a neural tube defect. (The best-known of these is spina bifida, in which the spinal cord is not completely enclosed.) Women at high risk, such as those who have already had a baby with this kind of defect, should take 4000 mcg (4 mg) daily, beginning before they become pregnant.
There is limited information on how much Qudexy XR or Trokendi XR is passed through breast milk. The potential for serious side effects in nursing infants is unknown. If you want to breastfeed your baby, check with your doctor about what seizure medicine would be best for you.
What are the effects of Topiramate XR on Seniors
Qudexy XR and Trokendi XR is often prescribed for people over 65 with epilepsy. Seniors have poorer kidney function than young adults, however, and Qudexy XR , Trokendi XR is eliminated from the body by the kidneys. So dosages of Qudexy XR and Trokendi XR for seniors should be smaller, to keep the medication from building up in the body.
Seniors also may be more sensitive to the side effects of Qudexy XR and Trokendi XR such as problems with their thinking.
What are the dose ranges for Topiramate XR?
The best amount is the amount that completely controls seizures without causing troublesome side effects. It depends on many factors, which are different for every individual. Follow the doctor's directions. Call if you have any questions.
In adults, Qudexy XR and Trokendi XR is usually started at 50 mg milligrams (mg) per day. Every week, the amount can be increased by 50-100 mg per day as required (if side effects are not troublesome) to a target dose of 200 to 400 mg per day in a once daily dose.. The recommended way to start and increase the dose is by starting at 50 mg per once per day. The second week, one takes 100 mg per day. The third week 150 mg per day. The fourth week 200 mg per day. The fifth week 300 mg per day and the sixth week 400 mg per day.
People over age 65 generally require a lower initial dose and particular caution with any increases.
Read the package insert of Topiramate XR
In the United States, companies that manufacture medicines are required to publish certain kinds of information about each product. This document is commonly known as a “package insert” because it is usually included with each package of the medicine.
You can also read these documents (also called "prescribing information") online. The U.S. package insert for Qudexy XR (topiramate) is found at:
The package insert for Trokendi XR can be found at:
Some of the information may differ in other countries.
To learn how to read and understand a package insert, see "How to read a package insert."

Primary Generalized Epilepsy
