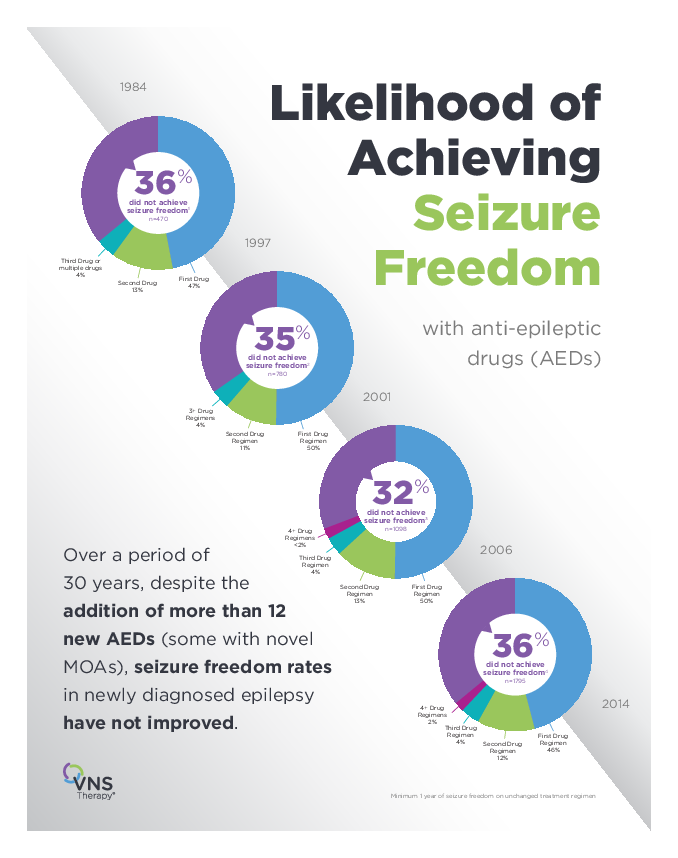
2020 Epilepsy Symposium

Meet our Public Symposium and Epilepsy Foundation Partners (Register Now)

Our pursuit of human health care (hhc)Eisai makes no mention of the word “pharmaceutical” in its company name. This is because what we really want to create is the everyday fulfillment of the precious hopes of patients and their families, which goes beyond the provision of medicines. “What are the patients' true needs ?” “What do families truly wish for ?” We want to get close to patients, see the situation from their perspectives and get a sense of their thoughts and feelings that cannot be expressed in words. We continue with our challenge to turn those thoughts and wishes into hopes for tomorrow. |
Eisai Resources |
|
|
Eisai Website |
Eisai Contact InformationPhone: 201-692-1100 Email: esi_medinfo@eisai.com |
|

|
|
VNS Resources
|
|
|
VNS Website |
VNS Contact InformationRyan Dixon, Utah Therapeutic Consultant cell (385) 352-6323, email Ryan.Dixon@livanova.com |
|

|
|
Intermountain Neurosciences Resources |
|
|
Intermountain Neurosciences Website |
Intermountain Neurosciences Contact Information |
|

|
Neurelis, Inc. is an innovation-driven neuroscience company providing a highly differentiated approach to target unmet medical needs. Neurelis is focused on the development and commercialization of product candidates for epilepsy and the broader central nervous system (CNS) market. We are built on a foundation of people with a passion for progress in serving the needs of people with epilepsy and those who care for them. Neurelis has reached a milestone in patient care with its first FDA-approved treatment. For further information, please visit our Product Page. For more information about Neurelis, please visit http://www.neurelis.com. |
|
|
|
Neurelis Website |
Neurelis Contact Information
|
|
.png)
|
BIOCODEX is a family-owned multinational pharmaceutical company founded in France in 1953, with proven expertise in treatments for the central nervous system. As the maker of DIACOMIT® (stiripentol), we are dedicated to providing education and support to healthcare providers, affected individuals, and their families. |
Biocodex ResourcesDIACOMIT can cause somnolence. Patients should be monitored for somnolence, and if it occurs, a dose reduction should be considered. Patients should be cautioned against engaging in hazardous activities requiring mental alertness, such as operating dangerous machinery or motor vehicles. DIOCOMIT can cause decreases in appetite and weight. The growth of pediatric patients should be carefully monitored. DIACOMIT can cause a significant decline in neutrophil and platelet count. Hematologic testing should be obtained prior to starting treatment and then every 6 months. DIACOMIT should generally be withdrawn gradually to minimize the risk of increased seizure frequency and status epilepticus. DIACOMIT powder for oral suspension contains phenylalanine which can be harmful to patients with phenylketonuria. DIACOMIT can increase the risk of suicidal thoughts or behavior in patients. Patients should be monitored for emergence or worsening of depression, suicidal thoughts or behavior, and/or any unusual changes in mood or behavior. The most common adverse reactions associated with DIACOMIT in clinical trials were somnolence (67%), decreased appetite (45%), agitation (27%), ataxia (27%), weight decreased (27%), hypotonia (24%), nausea (15%), tremor (15%), dysarthria (12%), and insomnia (12%). Please see full DIACOMIT Prescribing Information here.
|
|
|
Biocodex Websitewww.Diacomit.com |
Biocodex Contact Information |
|

|
|
Greenwich Biosciences Resources |
Follow us on Facebook |
Greenwich Biosciences WebsiteEpidiolex |
Greenwich Biosciences Contact Information |
|

|
Zogenix is a global biopharmaceutical company committed to developing transformative therapies to improve the lives of patients and their families living with rare diseases. |
Zogenix Resources |
Follow Us on Facebook |
Zogenix Website |
Zogenix Contact InformationHunter Liggett National Sales Director-West Mobile: +1 (206) 940-3604 Email: hliggett@zogenix.com |
|

|
Supernus Pharmaceuticals is specialty pharmaceutical company focused on developing and commercializing products for the treatment of central nervous system (CNS) diseases. Our extensive expertise in product development has been built over the past 25 years- fueling the success of more than 10 widely recognized products. We are dedicated to the goal of improving the treatment of CNS disorders, including epilepsy, attention-deficit/hyperactivity disorder ( ADHD), migraine and Parkinson’s disease.
|
Supernus Resourceshttps://www.oxtellarxr.com/co-pay-savings-program
|
Press Releases |
Supernus Website |
Supernus Contact Information |
|
Resources
Epilepsy Centers
Epilepsy centers provide you with a team of specialists to help you diagnose your epilepsy and explore treatment options.
Epilepsy Medication
Find in-depth information on anti-seizure medications so you know what to ask your doctor.
Epilepsy and Seizures 24/7 Helpline
Call our Epilepsy and Seizures 24/7 Helpline and talk with an epilepsy information specialist or submit a question online.
Tools & Resources
Get information, tips, and more to help you manage your epilepsy.